Ultraviolet Radiation
On this page
What is ultraviolet radiation?
Back to topUltraviolet (UV) radiation is similar to visible light in all physical aspects, except that it does not enable us to see things. The light that enables us to see things is referred to as visible light and is composed of the colours we see in a rainbow. The ultraviolet region starts right after the violet end of the rainbow.
In scientific terms, UV radiation is electromagnetic radiation just like visible light, radar signals and radio broadcast signals (see Figure 1). Electromagnetic radiation is transmitted in the form of waves. The waves can be described by their wavelength or frequency and their amplitude (the strength or intensity of the wave). Wavelength is the length of one complete wave cycle. For radiation in the UV region of the spectrum, wavelengths are measured in nanometers (nm), where 1 nm = one millionth of a millimetre.
Different wavelengths of electromagnetic radiation cause different types of effects on people. For example, gamma rays are used in cancer therapy to kill cancerous cells and infrared light can be used to keep you warm.
UV radiation has shorter wavelengths (higher frequencies) compared to visible light but has longer wavelengths (lower frequencies) compared to X-rays. UV radiation is divided into three wavelength ranges:
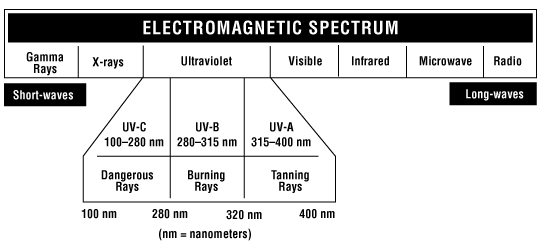
Figure 1 - Electromagnetic spectrum
Table 1 summarizes the general features of each type.
| Table 1 Types of Ultraviolet Radiation and Their Features | |
| Ultraviolet Radiation Type | General Features |
| Ultraviolet A radiation UVA, long-wave UV) |
|
| Ultraviolet B radiation (UVB, sunburn radiation) |
|
| Ultraviolet C radiation (UVC, short-wave UV) |
|
What are some sources of ultraviolet radiation?
Back to topSunlight is the greatest source of UV radiation. Man-made ultraviolet sources include several types of UV lamps, arc welding, and mercury vapour lamps.
UV radiation is widely used in industrial processes and in medical and dental practices for a variety of purposes, such as killing bacteria, creating fluorescent effects, curing inks and resins, light emitting diode (LED) plant grow lights, phototherapy and suntanning. Different UV wavelengths and intensities are used for different purposes.
Table 2 gives some examples of occupations with a potential risk of ultraviolet exposure. Table 3 gives examples of devices which employ UV radiation.
| Table 2 Workers at Potential Risk from Exposure to UV Radiation | |
|---|---|
Food and drink irradiators Salon workers and patrons Laboratory workers Lighting technicians Lithographic and printing workers Forensic experts Dentists and assistants Dermatologists and pediatricians General freight truckers | Outdoor workers Construction workers Contractors and surveyors Paint and resin curers Physiotherapists Plasma torch operators Welders Agriculture, forestry, fishing Greenhouse workers Photolithography |
| Table 3 Some Devices Emitting UV Radiation | |
|---|---|
Bactericidal lamps Black light lamps Carbon, xenon and other arcs Dental polymerizing equipment Fluorescence equipment Hydrogen and deuterium lamps Ultraviolet nail curing lamps | LED greenhouse lights Metal halide lamps Mercury lamps Plasma torches Phototherapy lamps Printing ink polymerizing equipment Welding equipment Counterfeit currency detectors |
What are some health effects of exposure to UV radiation?
Back to topSome UV exposure is essential for good health. It stimulates vitamin D production in the body. In medical practice, one example is UV lamps can be used for treating psoriasis (a condition causing itchy, scaly red patches on the skin).
Excessive exposure to ultraviolet radiation is associated with different types of skin cancer, sunburn, accelerated skin aging, as well as cataracts and other eye diseases. The severity of the effect depends on the wavelength (see Figure 2), intensity, and duration of exposure.
Effect on the skin
The shortwave UV radiation (UV-C) poses the maximum risk. The sun emits UV-C, but it is absorbed in the ozone layer of the atmosphere before reaching the Earth. Therefore, UV-C from the sun does not affect people. Some man-made UV sources also emit UV-C. However, the regulations concerning such sources restrict the UV-C intensity to a minimal level and may require the installation of special guards or shields and interlocks to prevent exposure to the UV.
The medium wave UV (UV-B) causes skin burns, erythema (reddening of the skin) and darkening of the skin. Prolonged exposures increase the risk of skin cancer.
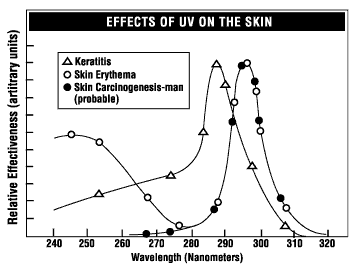
Figure 2 - Relative sensitivity of the eye and the
skin to UV radiation of different wavelengths.
Longwave UV radiation (UV-A) accounts for up to 95% of the UV radiation that reaches the Earth's surface. Although UV-A is less intense than UV-B, it is more prevalent and can penetrate deeper into the skin layers, affecting the connective tissue and blood vessels, which results in premature aging.
Certain chemicals and medications act as photosensitizing agents and enhance the effect of UV radiation from sunlight or other sources. Such agents include thiazide diuretics (drugs which cause excessive urine production), drugs used in the treatment of high blood pressure, certain antibiotics (tetracyclines, sulfonamides), cosmetics, and thiazine tranquillizers.
These are just a few examples; this is not intended to be a comprehensive list. However, it is important to know that these photosensitizing effects can occur in case people are exposed to UV radiation at work. For example, an inexperienced welder, who was taking a phenothiazine anti-depressant drug, suffered damage in both eyes in the part of the retina that absorbs short-wavelength light (bilateral maculopathy). He began complaining of eye problems a day after he was arc welding for two minutes without wearing any eye protection. This damage, which fortunately was reversible after several months, occurred because the drug he was taking sensitized him to the UV radiation to which he was exposed.
Various plants such as carrot, celery, dill, fig, lemon, and some types of weeds are known to cause photosensitivity. Exposure to fluids from these plants, especially if crushed, followed by exposure to sunlight, can cause dermatitis. Citrus fruit handlers and vegetable harvesters, gardeners, florists and bartenders are at risk for experiencing dermatitis following exposure to certain plants and then to sunlight (phytophotodermatitis).
Coal tar and creosote are examples of photosensitizing agents in the workplace.
Effects of repeated exposures (chronic effects) include skin aging and skin cancer. There is a strong causal link between skin cancer and prolonged exposure to solar UV and artificial sources.
Effect on the eyes
The eyes are particularly sensitive to UV radiation. Even a short exposure of a few seconds can result in a painful, but temporary condition known as photokeratitis and conjunctivitis. Photokeratitis is a painful condition caused by the inflammation of the cornea of the eye. The eyes water and vision is blurred. Conjunctivitis is the inflammation of the conjunctiva (the membrane that covers the inside of the eyelids and the sclera, the white part of the eyeball); (see Figure 3) which becomes swollen and produces a watery discharge. It causes discomfort rather than pain and does not usually affect vision.
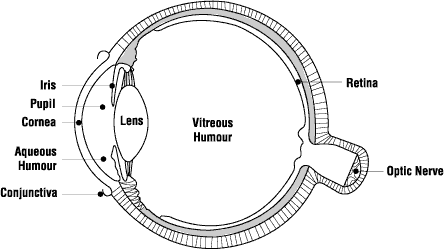
Figure 3 - The eye
Examples of eye disorders resulting from UV exposure include "flash burn", "ground-glass eye ball", "welder's flash" and "snow blindness" - depending on the source of the UV light causing the injury. The symptoms are pain, discomfort similar to the feeling of sand in the eye and an aversion to bright light.
The eyes are most sensitive to UV radiation from 210 nm to 320 nm (UV-C and UV-B). Maximum absorption by the cornea occurs around 280 nm. Absorption of UV-A in the lens may be a factor in producing cataract (a clouding of the lens in the eye).
How can you measure exposure?
Back to topThe intensity of UV radiation is measured in the units of milliwatts per square centimetre (mW/cm2) which is energy per square centimetre received per second. Also, it is measured in the units of millijoules per square centimetre (mJ/cm2), which is energy received per unit area in a given time.
A variety of instruments are commercially available for measuring UV radiation in the laboratory and in the workplace. Suppliers of workplace monitoring equipment can provide specifications and purchasing information.
Are there occupational exposure limits?
Back to topMany jurisdictions follow the limits recommended by the American Conference of Governmental Industrial Hygienists (ACGIH, 2025).
For the UV-A (315 to 400 nm), exposure to the eye should not exceed 1 milliwatt per square centimetre (1.0 mW/cm2) for periods greater than 1000 seconds (approximately 17 minutes). Additional exposure limits apply to the amount of UV light exposure to the skin and the eyes. The amount of UV exposure a person can receive on their skin or eyes during an 8-hour period varies with the wavelength of the UV radiation. For specifics or other limits, please consult the Ultraviolet Radiation section of the current edition of the ACGIH publication Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices®.
How do you protect yourself from UV radiation?
Back to topUV radiation is invisible and therefore does not stimulate the natural defences of the eyes. Workers must use eye and skin protection while working with UV radiation sources which present the potential for eye harmful exposure. The selection of eye protection depends on the type and intensity of the UV source. For more information, consult the CSA Standard CAN/CSA-Z94.3 2020 Eye and Face Protectors.
UV radiation is easily absorbed in a variety of materials. Shielding is usually easy to design. Mercury lamps and metal halide lamps have an outer glass cover to stop UV radiation, and are designed such that if the outer glass is broken, the lamp ceases to function.
For information on how to protect yourself from ultraviolet radiation in sunlight, please see the OSH Answers Skin Cancer and Sunlight.
- Fact sheet last revised: 2025-09-11

